They will position themselves around an atom so that there is as much space as possible between them. This applies regardless of the type of pairs they form.Įlectrons can form bonded pairs, lone pairs, double bonds, triple bonds, or exist as single unpaired electrons. The Valence shell electron-pair repulsion theory states that electrons will naturally repel each other. However, you can’t have more than two dots per side. In a Lewis structure, you can place the dots on any side of the symbol. If a shell is closed, the electrons that can be found in this shell won’t be shown on a Lewis electron dot diagram. It is common to omit the electrons that aren’t connected to the outer shells of an atom since they won’t form bonds with other elements and would only make Lewis structure diagrams more complicated. You would then add dots to represent the valence shell electrons connected to the atom.Ī valence electron is an electron located in the outer shell of the atom. You can create a Lewis structure electron dot diagram by simply writing the symbol for the atom you want to represent. You will often start with this simple visual representation called a Lewis structure to determine the correct three-dimensional structure for a molecule. This approach represents atoms and electrons in a two-dimensional manner. 
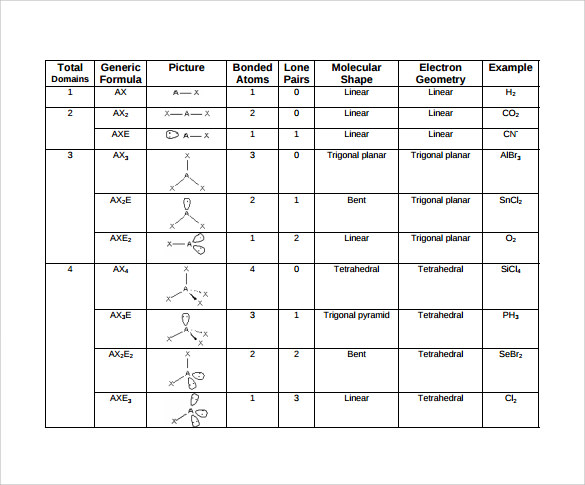
The Lewis theory is about how valence shell electrons bond with an atom. You can’t gain a thorough understanding of molecular geometry without studying the Lewis theory and the Valence-shell electron-pair repulsion theory. Lewis Theory And Valence-Shell Electron-Pair Repulsion Theory This results in ions being formed and is called an ionic bond. This means that the other atom loses all control of its electron, and the electronegative atom has complete control over both electrons. If one of the atoms is incredibly more electronegative than the other, this will result in the electron pair being dragged right over to that electronegative atom. At the same time, the opposing atom will become slightly positive. If one of the atoms is slightly more electronegative than the other, this will mean that one of the atoms has a fair share of electron density, and this results in that atom becoming slightly negative. To get a bond such as this, both atoms would usually be the same atom. If two atoms are equally electronegative, then both of those atoms have the same tendency to attract a pair of bonding electrons, and therefore it will be found on average halfway between the two atoms. What is electronegativity and why does it vary around a molecular geometry chart? Electronegativity is defined as the measure of the tendency of an atom to attract a bonding pair of electrons. This means that the two bonds cancel each other out, resulting in the molecule being non-polar. However, the molecular geometry of carbon dioxide is linear. For example, carbon dioxide has two polar bonds (C O). Not every molecule that has polar bonds is a polar molecule. Molecules that aren’t arranged in this way are called non-polar molecules. In this case, this type of molecule is called a polar molecule. The geometry of atoms in some molecules is arranged in such a way that one side has a negative charge and the other side has a positive electrical charge. Most students have an understanding of these structures thanks to the visual representations of molecules shown in class or textbooks even though they might not know about the rules of molecular geometry.Ĭertain molecules can be grouped as either polar or non-polar. You are probably already familiar with these structures if you have been studying chemistry for a while. These are the shapes and structures you will find in a molecular geometry chart: 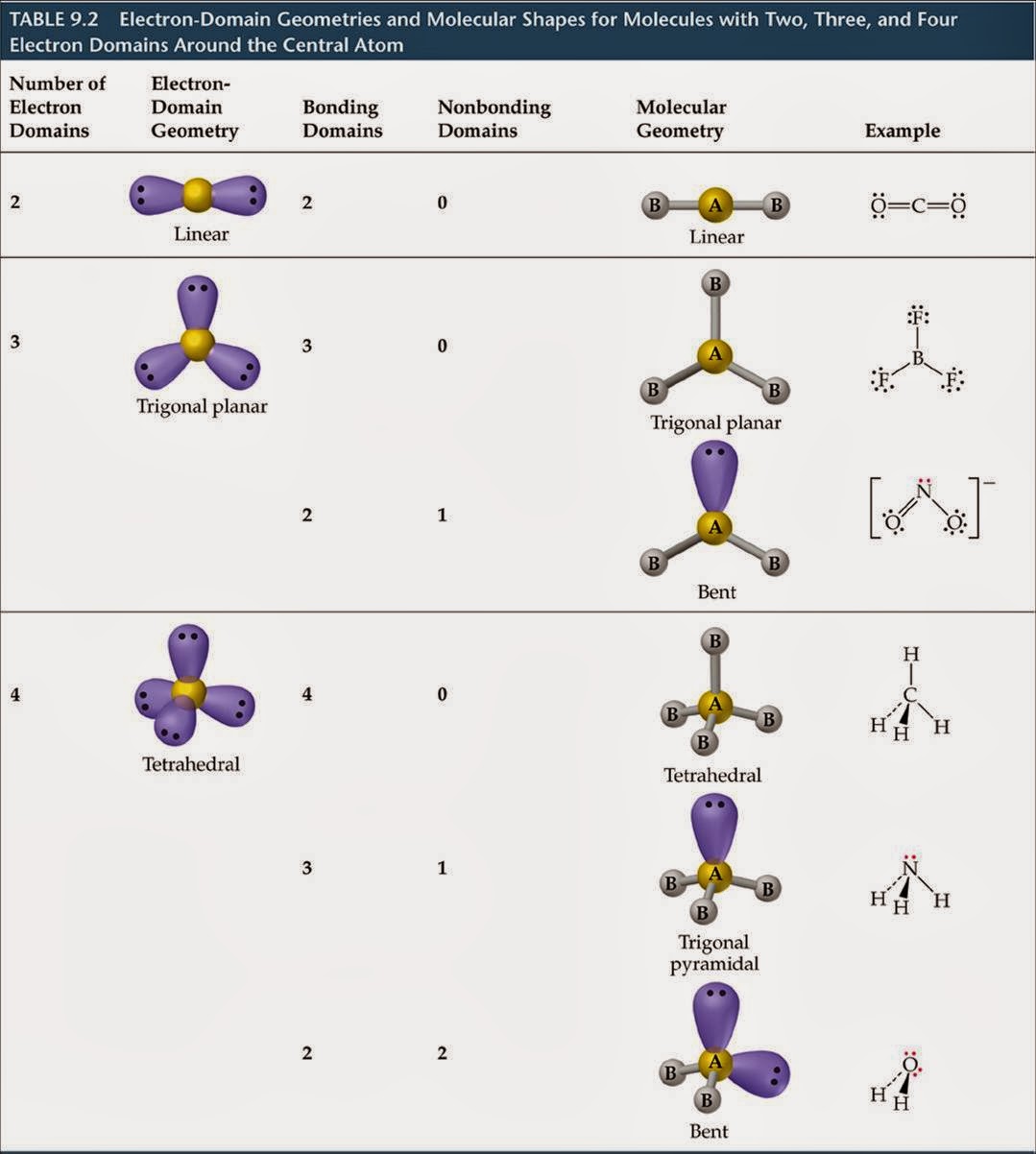
It is crucial to familiarize yourself with these common shapes so you can determine the correct one. Molecules obey certain laws when atoms and electrons connect with each other. Students will typically work with simple models at first before learning how to apply these concepts to create detailed models of more complex molecules. Molecular geometry can be applied regardless of how complex a molecule is. These representations can also help with other concepts, such as: These visual representations are interesting because they help students and scientists predict the shape, polarity, and biological activities of a molecule. Students and scientists can use these charts to create three-dimensional diagrams that represent molecules. A molecular geometry chart is a collection of rules on how molecules and electrons will connect and shape a molecule. Molecular geometry is the science of representing molecules in a three-dimensional manner. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
